BOXED WARNING: EMBRYO-FETAL TOXICITY
- Do not administer Letairis to a pregnant female because it may cause fetal harm. Letairis is very likely to produce serious birth defects if used by pregnant females, as this effect has been seen consistently when it is administered to animals
- Exclude pregnancy before the initiation of treatment with Letairis. Females of reproductive potential must use acceptable methods of contraception during treatment with Letairis and for one month after treatment. Obtain monthly pregnancy tests during treatment and 1 month after discontinuation of treatment
- Because of the risk of embryo-fetal toxicity, females can only receive Letairis through a restricted program called the Letairis REMS program
Contraindications
- Pregnancy: Letairis can cause fetal harm
- Idiopathic Pulmonary Fibrosis (IPF), including IPF patients with pulmonary hypertension (WHO Group 3)
Warnings and Precautions
- Embryo-fetal toxicity and Letairis REMS Program requirements:
- Prescribers must be certified with the program by enrolling in and completing training
- All female patients, regardless of reproductive potential, must enroll in the Letairis REMS Program
- Male patients are not enrolled in the program
- Pharmacies must be certified with the program and must dispense to female patients who are authorized to receive Letairis
Further information is available at www.letairisrems.com or 1-866-664-5327.
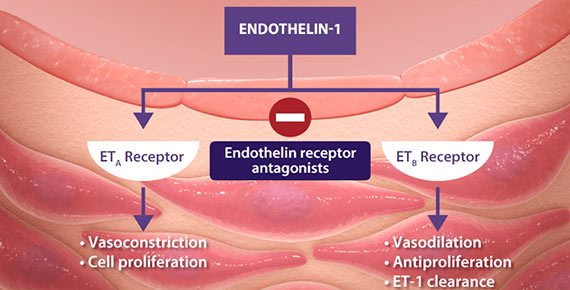
- Peripheral edema: Peripheral edema is a known class effect of endothelin receptor antagonists, and is also a clinical consequence of PAH and worsening PAH. Further evaluate patients who develop clinically significant fluid retention to determine the cause and possible need for edema treatment or to discontinue Letairis. In clinical studies, peripheral edema was more common with Letairis than with placebo (most edema was mild to moderate in severity); and with Letairis plus tadalafil than with either drug alone. There have also been postmarketing reports of fluid retention occurring within weeks after starting Letairis that required a diuretic, fluid management, or hospitalization for decompensating heart failure
- Pulmonary edema with pulmonary veno-occlusive disease (PVOD): Consider PVOD in patients who develop acute pulmonary edema during Letairis initiation and discontinue Letairis if PVOD is confirmed
- Decreased sperm counts have been observed in patients taking endothelin receptor antagonists and in animal fertility studies with ambrisentan. Counsel patients about potential effects on fertility
- Hematologic changes: Measure hemoglobin prior to initiation of Letairis, at 1 month, and periodically thereafter. Letairis initiation is not recommended for patients with clinically significant anemia. Consider discontinuing Letairis if clinically significant decreases in hemoglobin occur and other causes have been excluded. Decreases in hemoglobin and hematocrit have been observed within the first few weeks of Letairis treatment, which may persist during treatment. There have also been postmarketing reports of anemia requiring transfusion
Adverse Reactions
- Most common adverse reactions when used as monotherapy compared to placebo were peripheral edema (17% vs 11%), nasal congestion (6% vs 2%), sinusitis (3% vs 0%) and flushing (4% vs 1%)
- Most common adverse reactions in combination with tadalafil compared to Letairis or tadalafil monotherapy were peripheral edema (45% vs 38% or 28%), headache (41% vs 34% or 35%), nasal congestion (19% vs 16% or 11%), cough (18% vs 13% or 16%), anemia (15% vs 7% or 11%), dyspepsia (11% vs 3% or 12%), and bronchitis (10% vs 4% or 9%)
Drug Interactions
- Cyclosporine increases ambrisentan exposure by 2-fold, limit Letairis to 5 mg once daily
Use in Specific Populations
- Breastfeeding: Choose Letairis or breastfeeding
- Hepatic impairment: Letairis is not recommended in patients with moderate or severe hepatic impairment. Fully investigate cause of liver injury in patients who develop hepatic impairment; discontinue Letairis if liver aminotransferases are >5x ULN or if elevations are accompanied by bilirubin >2x ULN, or by signs or symptoms of liver dysfunction and other causes are excluded
Dosage and Administration
- Adult dosage: Initiate Letairis 5 mg once daily, with or without tadalafil 20 mg once daily. At 4-week intervals, consider either increasing to Letairis 10 mg or tadalafil 40 mg. Do not split, crush, or chew tablets
- Pregnancy testing: Initiate Letairis in females of reproductive potential only after a negative pregnancy test. Obtain monthly pregnancy tests during treatment
INDICATION
Letairis is indicated for the treatment of pulmonary arterial hypertension (PAH) (WHO Group 1) to improve exercise ability and delay clinical worsening; and in combination with tadalafil to reduce the risks of disease progression and hospitalization for worsening PAH, and to improve exercise ability. Studies establishing effectiveness included predominantly patients with WHO Functional Class II–III symptoms and etiologies of idiopathic or heritable PAH (60%) or PAH associated with connective tissue diseases (34%).
Letairis is indicated for the treatment of pulmonary arterial hypertension (PAH) (WHO Group 1) in combination with tadalafil to reduce the risks of disease progression and hospitalization for worsening PAH, and to improve exercise ability. The study establishing effectiveness included predominantly patients with WHO Functional Class II–III symptoms and etiologies of idiopathic or heritable PAH (58%) or PAH associated with connective tissue diseases (36%).1
Letairis is indicated for the treatment of pulmonary arterial hypertension (PAH) (WHO Group 1) to improve exercise ability and delay clinical worsening. Studies establishing effectiveness included predominantly patients with WHO Functional Class II–III symptoms and etiologies of idiopathic or heritable PAH (64%) or PAH associated with connective tissue diseases (32%).